The TIR Domain Signal Transduction Project
Toll-like receptors (TLRs) and interleukin-1 receptors (IL-1Rs) play crucial in many host immune responses. Signal transduction through these receptors ultimately leads to the activation of the NF-kB and AP-1 transcription factors.
The Toll/Interleukin-1 receptor (TIR) domain is the conserved intracellular domain of the two families of receptors. This domain is also shared by the downstream adapter molecule MyD88. Upon receptor activation, it is believed that a TIR domain signaling complex is formed between the receptor and the adapter TIR domains. A single point mutation in the TIR domain of murine TLR4 (the Lps_d mutation) renders the mice hyporesponsive to lipopolysaccharide (LPS).
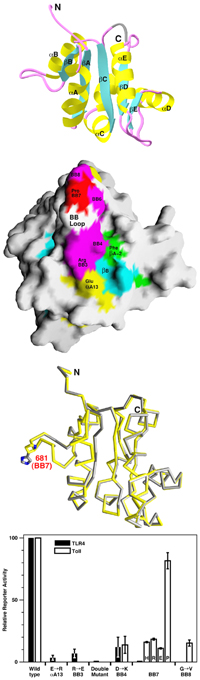
Major findings from this project- The structures of the TIR domains of human TLR1 and TLR2 have been determined.
- A conserved surface patch on the TIR domain, corresponding to the BB loop, may be important for receptor signaling.
- Functional studies using receptor reporter assays with Drosophila Toll and human TLR4 confirms the importance of the conserved surface patch.
- The Lps_d mutation does not disrupt the structure of the domain. It is likely that it disrupts the formation of the TIR domain complex.
- The structure of the TIR domain of human IL-1RAPL has been determined.
Publications from this project
- Y. Xu, X. Tao, B. Shen, R. Medzhitov, J.L. Manley & L. Tong. (2000). Structural basis for signal transduction by the Toll/interleukin-1 receptor domain. Nature, 408, 111-115. Medline | Reprint(PDF)
- X. Tao, Y. Xu, Y. Zheng, A. Beg & L. Tong. (2002). An extensively-associated dimer in the structure of the C713S mutant of the TIR domain of human TLR2. Biochem. Biophys. Res. Commun. 299, 216-221.
- J. Khan, E.K. Brint, L.A.J. O'Neill & L. Tong. (2004). Crystal structure of the Toll/interleukin-1 receptor (TIR) domain of IL-1RAPL. J. Biol. Chem. 279, 31664-31670.
- L. Tong. (2005). Toll/interleukin-1 receptors and innate immunity. in G. Waksman, M.G. Caparon & S. Hultgren, eds. Structural basis of bacterial pathogenesis. pp. 241-263. ASM Press, Washington, DC.
- X. Tao & L. Tong. (2009). Expression, purification, and crystallization of toll/interleukin-1 receptor (TIR) domains. Methods Mol. Biol. 517, 81-88. Reprint(PDF)
Funding for this project
- NIH R01 AI49475 (2001-2006)
© copyright 2000-2017, Liang Tong.