The p38 MAP Kinase Project
The p38 MAP kinase is activated in response to cellular stress, such as stimulation by LPS. A series of compounds, known as pyridinyl-imidazoles, can inhibit the p38-mediated production of pro-inflammatory cytokines (TNF and IL-1) in response to LPS stimulation. These compounds are highly specific against the p38 MAP kinase.
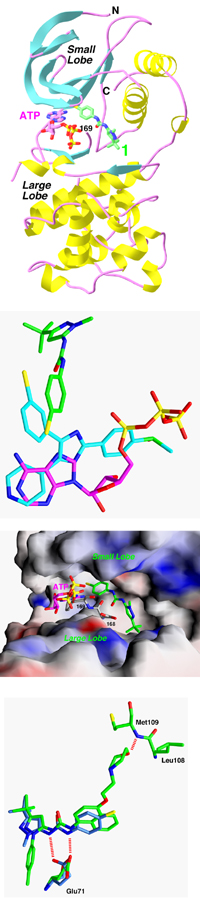
Major findings from this project- The pyridinyl-imidazole inhibitors bind in the ATP pocket, and is therefore competitive against ATP.
- The specificity of this series of compounds is determined by the presence of the 4-phenyl ring, which occupies a binding pocket in the kinase that is not utilized by ATP.
- A threonine residue in p38 MAP kinase is crucial for the presence of this pocket. In most other kinases, this residue is replaced by larger Gln or Met residues. The T->Q or T->M single-point mutants of p38 are insensitive to the inhibitors.
- A new, diaryl urea series of highly selective p38 MAP kinase inhibitors has been developed.
- The compound BIRB796 has Ki of 0.1 nM, with a very slow off rate.
- The compound binds to an allosteric pocket in p38, which is revealed when the highly conserved DFG motif assumes a different conformation (the DFG-out conformation).
- The compounds are not directly competitive with ATP binding. But the DFG-out conformation is not compatible with ATP binding.
Publications from this project
- S. Pav, D. M. White, S. Rogers, K. M. Crane, C. L. Cywin, W. Davidson, J. Hopkins, M. L. Brown, C. A. Pargellis & L. Tong. (1997). Crystallization and preliminary crystallographic analysis of recombinant human p38 MAP kinase. Protein Science, 6, 242-245. Reprint(PDF)
- L. Tong, S. Pav, D. M. White, S. Rogers, K. M. Crane, C. L. Cywin, M. L. Brown & C. A. Pargellis. (1997). A highly specific inhibitor of human p38 MAP kinase binds in the ATP pocket. Nature Struct. Biol. 4, 311-316. Reprint(PDF)
- C. Pargellis, L. Tong, L. Churchill, P.F. Cirillo, T. Gilmore, A.G. Graham, P.M. Grob, E.R. Hickey, N. Moss, S. Pav & J. Regan. (2002). Inhibition of p38 MAP kinase by utilizing a novel allosteric binding site. Nature Struct. Biol. 9, 268-272. Reprint(PDF)
- J. Regan, S. Breitfelder, P. Cirillo, T. Gilmore, A.G. Graham, E. Hickey, B. Klaus, J. Madwed, M. Moriak, N. Moss, C. Pargellis, S. Pav, A. Proto, A. Swanimer, L. Tong & C. Torcellini. (2002). Pyrazole urea-based inhibitors of p38 MAP kinase: From lead compound to clinical candidate. J. Med. Chem. 45, 2994-3008. Reprint(PDF)
© copyright 2000-2017, Liang Tong.