The Separase Project
Updated Jan. 2021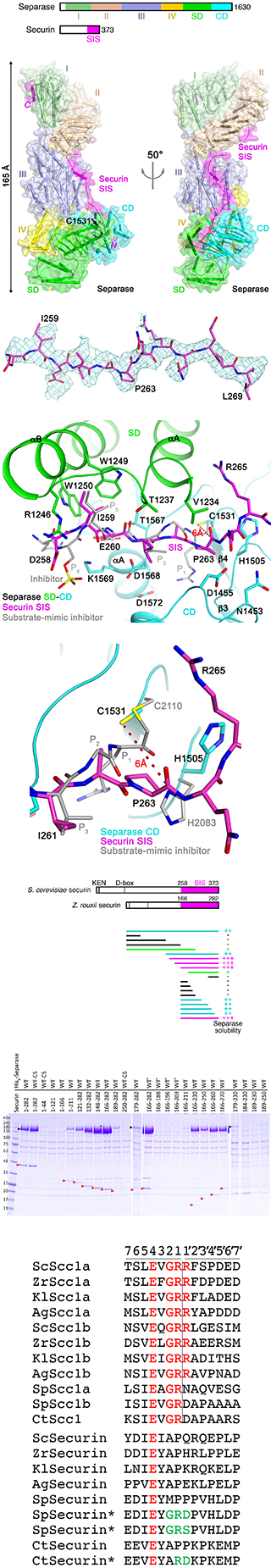
Separase is a large (140-250 kD) eukaryotic endopeptidase belonging to the CD clan of cysteine proteases, which also includes caspases and gingipain. It has essential roles in chromosome segregation, by cleaving the kleisin subunit (Scc1/Rad21/Mcd1 for mitosis and Rec8 for meiosis) of the cohesin complex that entraps sister chromatids during cell division. It also has important functions in other cellular events, such as stabilizing the anaphase spindle by cleaving and localizing the kinetochore-associated protein Slk19, and regulating centriole disengagement in mammals by cleaving pericentrin/kendrin. Overexpression of separase is linked to aneuploidy and tumorigenesis, making it a potential target for drug discovery.
The activity of separase is tightly regulated. Securin, a natively unfolded protein in solution, acts as both a chaperone and a potent inhibitor of separase.
The primary structure of separase contains a C-terminal caspase-like catalytic domain (CD) of ~200 residues and an N-terminal a-helical regulatory region, which is poorly conserved. Securin has a KEN-box and a D-box in its N-terminal region which are crucial for ubiquitination by APC/C, while its C-terminal region mediates the binding and inhibition of separase.
Major findings from this project- The crystal structure of the yeast separase-securin complex has been determined at up to 2.6 A resolution.
- The separase-securin complex assumes a highly elongated structure.
- Residues 258-373 of securin, named the separase interaction segment (SIS), is primarily in an extended conformation and traverses the entire length of separase.
- Residues 258-269 of securin are located in the separase active site, inhibiting its activity by blocking substrate binding. Securin itself is not cleaved as its P1 residue assumes a different conformation and also clashes with the second member of the catalytic dyad.
- Contacts between securin and separase outside the separase active site are crucial for stabilizing the complex.
Publications from this project
- S. Luo & L. Tong. (2017). Molecular mechanism for the regulation of yeast separase by securin. Nature, 542, 255-259.
- S. Luo & L. Tong. (2018). Structural biology of the separase-securin complex with crucial roles in chromosome segregation. Curr. Opin. Struct. Biol. 49, 114-122.
- S. Luo & L. Tong. (2020). Structure and function of the separase-securin complex. in Macromolecular Protein Complexes III: Structure and Function, Chapter 4, pp. 217-232. J.R. Harris & J. Marles-Wright Eds. Springer, New York, Heidelberg, Dordrecht, London. Alternatively Subcellular Biochem. 96, 217-232.
Funding for this project
- NIH including R35GM118093
© copyright 2019-2021, Liang Tong.