
The ATP-citrate Lyase (ACLY) Project
Updated Jan. 2020
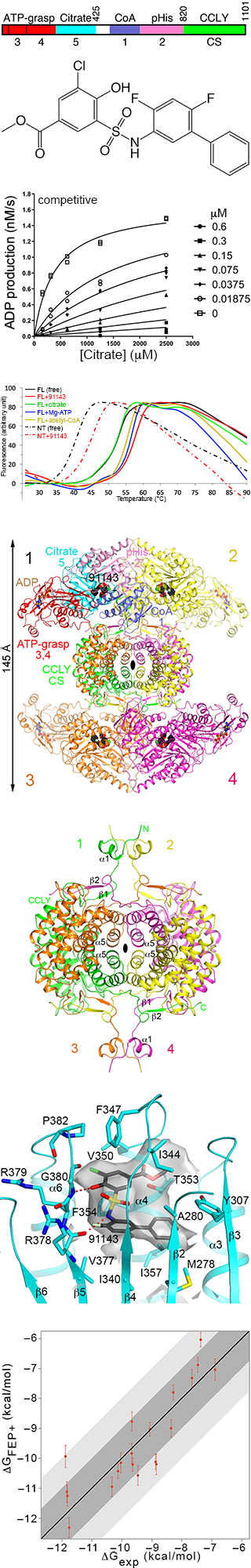
ATP-citrate lyase (ACLY) catalyzes the ATP-dependent conversion of citrate and CoA to
oxaloacetate and acetyl-CoA. It is one of the major sources of cytosolic
acetyl-CoA, and is a central metabolic enzyme. The acetyl-CoA product is crucial
for fatty acid metabolism, cholesterol biosynthesis, and post-translational modification
of proteins (acetylation and prenylaion).
ACLY activity is required for proliferation by many tumors, making it an attractive
target for drug discovery. ACLY is also a target against dyslipidemia and hepatic
steatosis. A compound is in phase III clinical trials against dyslipidemia. The currently
known inhibitors of ACLY all have weak activity, with IC50 greater than 100 uM.
ACLY is a homotetramer of 500 kD, wich each monomer containing ~1100 residues.
The reaction proceeds in four steps, with the formation of a citryl-CoA intermediate.
Crystal structure information is available for the first ~820 residues of the enzyme, but
this N-terminal fragment is monomeric and cannot catalyze the overall reaction. The C-terminal
~300 residues have weak homology to citrate synthase (CS).
Major findings from this project
- The structure of human ACLY homotetramer has been determined by cryo-EM.
- The C-terminal domain of ACLY forms the central core of the tetramer,
showing extensive interactions among the four monomers. The N-terminal segment
(~820 residues) is projected away from the core, showing only small interactions
among them.
- The C-terminal domain also contains an active site, but it does not have
CS activity as a crucial His residue in the active site of CS
is a Phe in ACLY. Instead, the C-terminal domain has citryl-CoA lyase (CCLY)
activity.
- A series of inhibitors with low nanomolar affinity for ACLY has been
developed. It is competitive versus the citrate substrate, but noncompetitive
versus the other substrates.
- The compound NDI-091143 is bound to the citrate domain, but not in the active site.
Intead, the compound is bound in an allosteric cavity next to the active site, in the
hydrophobic core of the citrate domain.
- Extensive conformational changes in the citrate domain are needed for
the formation of this binding site for the compound. These changes disrupte the citrate
binging site, thereby explaining why the compound is competitive against citrate.
- The binding mode of the compound is supported by free-energy pertubation
calculations on 19 other analogs, showing excellent correlation between predicted
binding affinity and observed activity.
- This allosteric cavity enhances the druggability of ACLY, and is
likely more amenable for developing ACLY inhibitors.
Publications from this project
-
J. Wei, S. Leit, J. Kuai, E. Therrien, S. Rafi,
H.J. Harwood Jr, B. DeLaBarre & L. Tong. (2019).
An allosteric mechanism for potent inhibition of human ATP-citrate lyase.
Nature, 568, 566-570.
-
J. Puccini, J. Wei, L. Tong & D. Bar-Sagi. (2023). Cytoskeletal association of ATP citrate lyase controls the mechanodynamics of macropinocytosis. Proc. Natl. Acad. Sci. USA, 120, e2213272120.
Funding for this project
© copyright 2019-2026, Liang Tong.


