
The AMP-activated Protein Kinase (AMPK/SNF1) Project
AMP-activated protein kinase (AMPK) is a master metabolic regulator. AMPK activation
leads to the down regulation of energy-demanding, anabolic processes (fatty acid
and cholesterol biosynthesis, gluconeogenesis, glycogen synthesis and others) and the
stimulation of energy-producing, catabolic processes (fatty acid oxidation, glucose
uptake, glycolysis and others). AMPK is a dual inhibitor of ACC1 and ACC2, by
phosphorylating them near the N terminus.
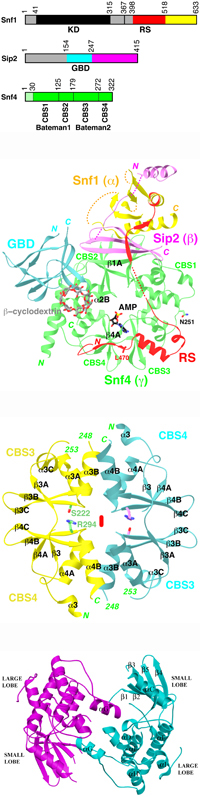
AMPKs are hetero-trimeric enzymes, with a catalytic alpha subunit and two
regulatory (beta and gamma) subunits.
The alpha subunit contains the protein kinase domain,
and the beta subunit a glycogen binding domain. The gamma subunit contains 4
repeats of the cystathionine-beta-synthase (CBS) motif. Each CBS tandem pair is
also known as the Bateman domain.
AMP is believed to regulate AMPK by
binding to the gamma subunit.
AMPK is an attractive target for drug discovery. Metformin is believed to function
through indirect activation of this kinase.
Major findings from this project
- The structure of the heterotrimer core of yeast AMPK
homolog SNF1 has been determined at 2.6 A resolution.
- The regulatory segment (RS) in the alpha subunit (Snf1)
is sequestered by the gamma subunit (Snf4) in the structure.
- The glycogen binding domain (GBD) is located near Snf4
in the structure.
- The structure of the catalytic domain of the alpha subunit
of yeast AMPK (Snf1) has been determined at 2.2 A resolution.
- A dimer of the kinase domain was observed in the crystal.
The biological relevance of this dimer remains to be
determined.
- The structure of the second Bateman domain of yeast
Snf4 (the gamma subunit) has been determined at
1.9 A resolution.
- The Bateman2 domain alone forms a dimer with an extensive
interface, with a prominent pocket in the center of the dimer.
Publications from this project
-
M.J. Rudolph, G.A. Amodeo, Y. Bai & L. Tong. (2005). Crystal
structure of the protein kinase domain of yeast AMP-activated
protein kinase Snf1. Biochem. Biophys. Res. Commun.
337, 1224-1228.
Reprint(PDF)
-
M.J. Rudolph, G.A. Amodeo, S.H. Iram, S.-P. Hong, G. Pirino, M.
Carlson & L. Tong. (2007). Crystal structure of the Bateman2
domain of yeast Snf4: dimeric association and relevance for
AMP binding. Structure, 15, 65-74.
Reprint(PDF)
-
G.A. Amodeo, M.J. Rudolph & L. Tong. (2007). Crystal structure of the
heterotrimer core of Saccharomyces cerevisiae AMPK homolog SNF1.
Nature, 449, 492-495.
Reprint(PDF)
-
G.A. Amodeo, M. Momcilovic, M. Carlson & L. Tong. (2010).
Biochemical and functional studies on the regulation of the
Saccharomyces cerevisiae AMPK homolog SNF1.
Biochem. Biophys. Res. Commun., 397, 197-201.
Reprint(PDF)
-
M.J. Rudolph, G.A. Amodeo & L. Tong. (2010). An inhibited
conformation for the protein kinase domain of the Saccharomyces
cerevisiae AMPK homolog Snf1.
Acta Cryst., F66, 999-1002.
Reprint(PDF)
Funding for this project
© copyright 2006-2026, Liang Tong.


