
The BEACH Domain Project
BEACH domains are highly conserved in a large family of eukaryotic proteins that
are involved in vesicle trafficking, membrane dynamics, cytokinesis, and receptor
signaling.
The BEACH domain was first identified in the proteins beige and
CHS. Chediak-Higashi Syndrome (CHS) is a rare, autosomal recessive
disorder that can cause severe immunodeficiency and albinism in humans. Unless
treated by bone marrow transplantation, CHS patients generally die in childhood.
beige is the name for CHS disease in mice and other animals.
BEACH domains share no detectable sequence homology with other proteins in the database.
Its exact biochemical/biological function is currently not known. BEACH-containing
proteins are generally very large, from 900 residues for the protein FAN to 3800 residues
for CHS.
Major findings from this project
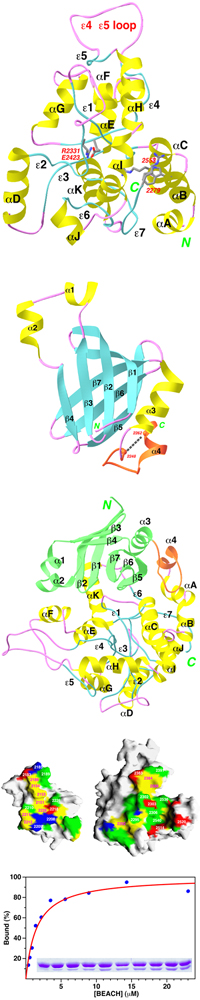
- The crystal structure of the BEACH domain of human neurobeachin has been
determined at 2.9A resolution.
- The BEACH domain has a novel and unusual fold. Several polypeptide segments
in the core of the domain do not assume regular secondary structures.
-
A novel PH domain just prior to the BEACH domain in the amino acid sequence
was revealed from the structural studies.
-
There is an extensive interface between the PH and BEACH domains, and the
affinity between the two domains have been estimated to be around 1 uM
based on GST pull-down experiments.
-
The BEACH and PH domains may function as a single unit.
Publications from this project
-
G. Jogl, Y. Shen, D. Gebauer, J. Li, K. Wiegmann, H. Kashkar, M. Kronke & L. Tong. (2002).
Crystal structure of the BEACH domain reveals an unusual fold and extensive association with a
novel PH domain. EMBO J. 21, 4785-4795.
-
D. Gebauer,* J. Li,* G. Jogl, Y. Shen, D.G. Myszka & L. Tong.
(2004). Crystal structure of the PH-BEACH domains of human
LRBA/BGL. Biochem. 43, 14873-14880. (*-equal first authors)
Funding for this project
© copyright 2003-2026, Liang Tong.


