
The Propionyl-CoA Carboxylase (PCC) Project
Propionyl-CoA carboxylase (PCC) catalyzes the biotin-dependent carboxylation
of propionyl-CoA to produce D-methylmalonyl-CoA. PCC is
essential for the catabolism of the amino acids Thr, Val, Ile and Met,
cholesterol, and fatty acids with an odd number of carbon atoms.
Deficiencies of PCC activity in humans are linked to the disease
propionic acidemia (PA), an autosomal recessive disorder that
can be fatal in infants.
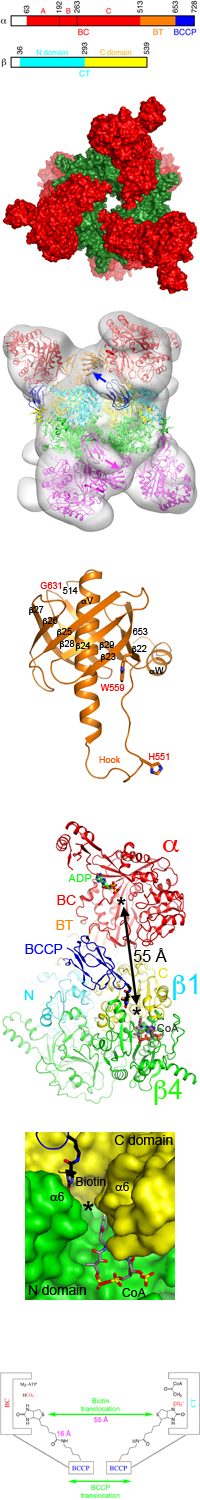
PCC holoenzyme is a 750-kD a6b6 dodecamer.
The a subunit is known to contain the biotin carboxylase (BC) and biotin
carboxyl carrier protein (BCCP) domains, and the b subunit
supplies the carboxyltransferase (CT) activity.
Major findings from this project
- The crystal structure of the a6b6 holoenzyme of a bacterial PCC
chimera (RpPCCa-RdPCCb) has been
determined at 3.2A resolution.
- The human PCC holoenzyme has the same structure, based on
cryo-EM reconstruction at 15A resolution.
- The holoenzyme has 32 symmetry, and
contains a central b6 hexameric core, in the shape
of a short cylinder with a small hole along its axis.
- The a subunits are arranged as monomers in the holoenzyme, on both
ends of the b6 core.
- Interactions between the a and b subunits are mediated primarily by the BT
domain in the a subunit
(named as it mediates BC-CT interactions),
located between the BC and BCCP domains in the primary
sequence and
previously unrecognized by sequence analysis.
- The BT domain
has a novel fold,
with a long, central helix surrounded by an eight-stranded anti-parallel b-barrel.
The structure has very weak similarity to that of the PT domain in the structure
of pyruvate carboxylase (PC), which has a helix surrounded by a four-stranded
anti-parallel b-sheet.
- The active sites of BC and CT are separated by 55A in the holoenzyme, indicating
that the entire BCCP domain must translocate during catalysis.
- BCCP and its biotin are located in the CT active site in the structure, providing
insight into their involvement in the carboxyltransfer reaction.
- The structure of the holoenzyme provides a molecular basis for understanding the
large collection of disease causing mutations in PCC.
Publications from this project
-
C.S. Huang*, K. Sadre-Bazzaz*, Y. Shen, B. Deng, Z.H. Zhou & L. Tong. (2010).
Crystal structure of the a6b6 holoenzyme of propionyl-coenzyme A
carboxylase.
Nature, 466, 1001-1005. (*-equal first authors)
Reprint(PDF)
Funding for this project
© copyright 2010-2026, Liang Tong.


